
Recently, the top-tier international journal Nature published the findings of a multicenter clinical study on OTOF gene therapy, led by Professor Shu Yilai from the Eye & ENT Hospital of Fudan University, with participation from Associate Professor Xiong Yuanping (Deputy Director of the Department of Otolaryngology Head and Neck Surgery at the First Affiliated Hospital of Nanchang University). As one of the eight core centers nationwide, the First Affiliated Hospital of Nanchang University actively participated in this landmark study, and completed participant recruitment, clinical intervention, standardized follow-up, and data collection with high quality. This work provided key clinical support for verifying the safety and efficacy of the gene therapy, helping to advance congenital deafness treatment toward a new stage of clinical application.
The study, titled “Multicentre gene therapy for OTOF-related deafness followed up to 2.5 years”, reported multicentre follow-up results over a period of up to 2.5 years for an innovative gene therapy targeting DFNB9 congenital deafness caused by OTOF gene mutations, and identified core biomarkers predictive of therapeutic efficacy. As the world’s first clinical trial of gene therapy for congenital deafness, this study provides clinical evidence involving the largest number of participants, the longest follow-up duration, and the broadest age coverage to date. It systematically confirms the safety and efficacy of gene therapy across all age groups, marking a crucial leap from case-by-case breakthroughs to multicentre validation and widely accessible clinical treatment, and bringing hope for hearing recovery to patients with OTOF-related deafness worldwide.
Hearing Restored in 90% of Participants, Safe and Effective Across All Ages

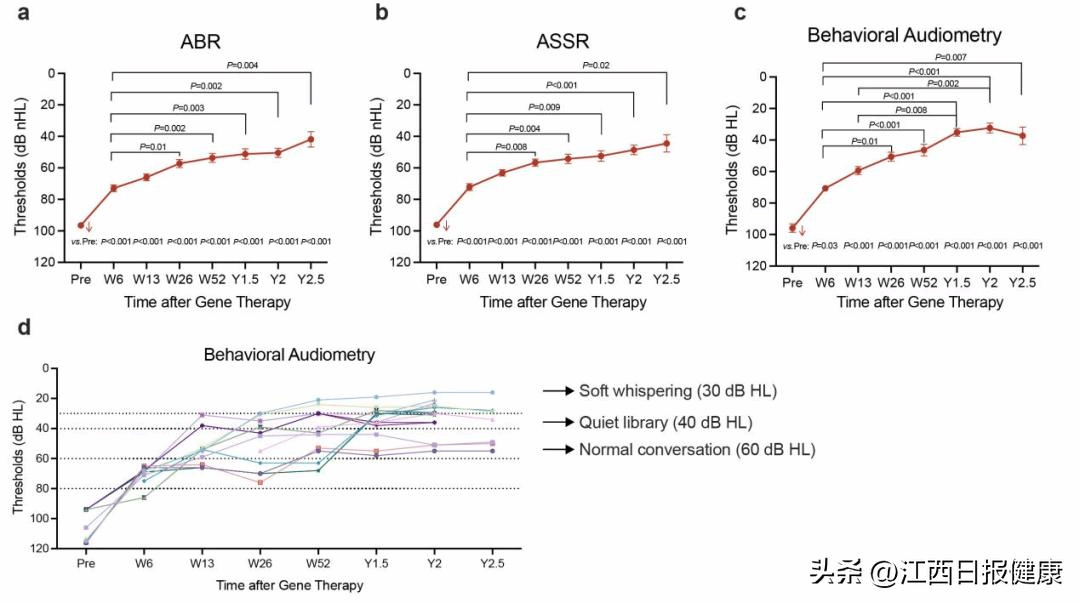
(Average hearing thresholds over time in participants with hearing recovery. Arithmetic mean thresholds of ABR (a), ASSR (b), and behavioral audiometry (c) at 0.5, 1, 2, and 4 kHz in participants from baseline to 2.5 years. d, the average hearing threshold of behavioral audiometry from baseline to 2 years in 15 ears and 2.5 years in 7 ears, respectively.)
The study results showed that AAV-hOTOF gene therapy led to clinically meaningful hearing recovery in 90% of participants, with a gradual and stable improvement in hearing. The mean auditory brainstem response (ABR) threshold improved progressively from >97 dB nHL before treatment to 42 dB nHL at 2.5 years post-treatment. The mean auditory steady-state response (ASSR) threshold improved from >96 dB nHL before treatment to 44 dB nHL at 2.5 years post-treatment, and the mean behavioral audiometry threshold improved from >96 dB HL before treatment to 37 dB HL at 2.5 years post-treatment. In long-term follow-up, all participants who completed the 2.5-year follow-up could recognize conversational speech; 57% could identify library-quiet sounds, and 43% could hear a whisper. Along with hearing recovery, participants showed continuous improvement in hearing and speech abilities, with further enhancement in speech perception in noisy environments, enabling them to better integrate into social life, study, and daily activities. This therapy was also effective in adult participants, with two out of three achieving hearing improvement, confirming the value of gene therapy for adult patients with deafness.
First Identification of Core Factors Influencing the Efficacy of OTOF Gene Therapy, Guiding Precision Diagnosis and Treatment
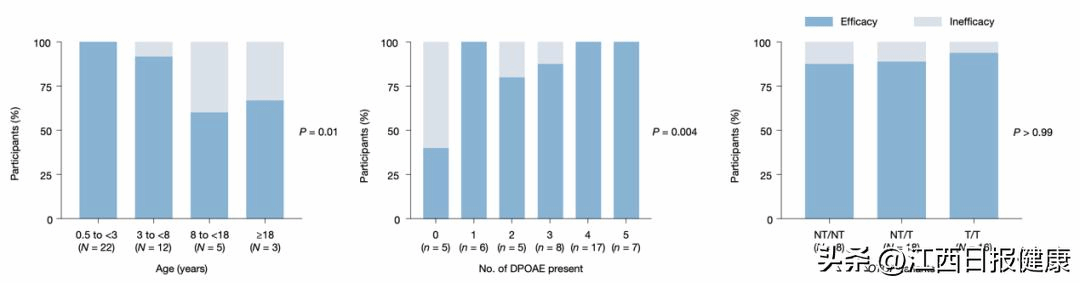
Distribution of treatment outcomes (efficacy and inefficacy) by baseline characteristics, including age group, number of present DPOAEs at baseline and OTOF variants, using two-tailed Fisher’s exact test.N, number of participants; n, number of treated ears.
Through in-depth analysis, the research team explored the factors influencing the treatment outcomes of OTOF gene therapy, which included age, the number of present distortion product otoacoustic emissions (DPOAEs) at baseline, and OTOF mutation types. The study found that participants aged 0.5 to 18 years showed greater treatment efficacy and degree of hearing improvement compared to those older than 18 years. Among them, the efficacy rate was 100% in young children aged 0.5 to under 3 years, 92% in children aged 3 to under 8 years, and 60% in adolescents aged 8 to under 18 years. The number of present DPOAEs at baseline can predict treatment outcomes. As an important measure for assessing cochlear outer hair cell function, DPOAEs reflect the integrity of the sensory cells essential for hearing. The study found that participants with more present DPOAEs at baseline achieved better hearing recovery after treatment, a finding that promises to serve as an important biomarker for predicting the efficacy of OTOF gene therapy. Participants harboring biallelic non-truncating variants (NT/NT) had better behavioral audiometry thresholds after treatment compared to those harboring truncating variants (NT/T, T/T), but there was no significant difference in efficacy rates among different mutation types (88%–94%), indicating that this therapy is applicable to all types of OTOF gene mutations.
Gene therapy for deafness is a major breakthrough in medicine, fully demonstrating the innovative research and clinical trial capabilities of Chinese scholars. This project has been recognized as one of the “Top 10 Global Clinical Breakthroughs of 2024” by Yixuejie (a leading Chinese medical media platform) and as a “Major Medical Advance in China 2024” by the Chinese Academy of Medical Sciences. Under the guidance of Professor Jiang Hongqun, Chief Expert of Otolaryngology Head and Neck Surgery at the hospital, Associate Professor Xiong Yuanping’s team is dedicated to precision diagnosis and treatment of deafness. As one of the core sub-centers in this multicentre study, the team pioneered the OTOF gene therapy program for deafness within the province, reflecting the hospital’s comprehensive strength in the fields of otology, audiology, and the clinical translation of gene therapy. In the future, the First Affiliated Hospital of Nanchang University will continue to collaborate deeply with all centers to further advance the clinical translation and application of gene therapy for deafness, allowing precision medicine to benefit more patients with hearing loss.
Associate Professor Xiong Yuanping from the First Affiliated Hospital of Nanchang University, together with Professor Shu Yilai, Professor Li Huawei, and others from the Eye & ENT Hospital of Fudan University, served as co-corresponding authors for this study. Professor Jiang Hongqun, Attending Physician Wang Jun, and Attending Physician Guan Lina from the Department of Otolaryngology Head and Neck Surgery at the First Affiliated Hospital of Nanchang University, as core research members, made significant contributions to the implementation and outcomes of this research.




